Stephen Osmani
Professor
804 Riffe Building
496 West 12th Avenue
Columbus, OH
43210-1292
Areas of Expertise
- Cell Biology of Fungi
- Aspergillus nidulans Biofilms
- Gaseous Microenvironments
My laboratory is conducting research aimed at elucidating how the cell cycle is regulated with focus on the regulation of mitosis, the universal mechanism by which our DNA is segregated equally to daughter cells during growth and development. We utilize the model filamentous fungus Aspergillus nidulans in these studies. This enables us to use the power of classical genetics (including the parasexual cycle and heterokaryon rescue), molecular genetics, cell biology, genomics/proteomics, and biochemistry to identify and study novel proteins involved in cell cycle regulation. These findings will provide insights into how mitosis is regulated in filamentous fungi, a poorly understood group of organisms which have far-reaching beneficial and detrimental impacts on mankind. The findings should also provide insights into how mitosis is regulated in higher eukaryotes with implications for how cancer develops and can be treated.
Recent work in the lab has focused on understanding how nuclear transport is regulated during mitosis. In higher eukaryotes the nuclear envelope is dismantled during mitosis (open mitosis) by ill-defined mechanisms. However in fungi, such as A. nidulans, mitosis occurs within intact nuclei (closed mitosis). During closed mitosis, proteins such as tubulin which forms the mitotic spindle enter nuclei only during mitosis. It had long been assumed that specific transport pathways through nuclear pore complexes 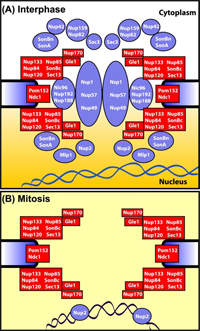 (NPCs, the structures providing regulated conduits across the nuclear envelope between the cytoplasm and the nucleoplasm) were modified during closed mitosis. However, our recent findings have demonstrated that in A. nidulans NPCs are opened to allow diffusion in and out of nuclei during mitosis rather than specific transport pathways being modified. This is achieved by the release of over half of the ~30 NPC proteins from NPCs specifically during mitosis (See Figure, taken from Osmani et al., Molecular Biology of the Cell 2006).
(NPCs, the structures providing regulated conduits across the nuclear envelope between the cytoplasm and the nucleoplasm) were modified during closed mitosis. However, our recent findings have demonstrated that in A. nidulans NPCs are opened to allow diffusion in and out of nuclei during mitosis rather than specific transport pathways being modified. This is achieved by the release of over half of the ~30 NPC proteins from NPCs specifically during mitosis (See Figure, taken from Osmani et al., Molecular Biology of the Cell 2006).
The NPC proteins that remain form a core conduit between the inside and outside of nuclei. During exit from mitosis the dispersed NPC proteins return back to the core NPC structure and reestablish regulated transport. These findings provide a new paradigm for how mitosis can be regulated and indicate A. nidulans mitosis is an intermediate in complexity between closed and open mitoses.
In addition to explaining how nuclear transport is regulated during A. nidulans mitosis these new findings provide a framework to help decipher how the massive NPC structure is first dismantled then reassembled during mitosis. We know that protein phosphorylation plays a critical role and we have shown that the NIMA and Cdk1 mitotic kinases promote disassembly of the NPC. The challenge now is to define which NPC proteins are phosphorylated then dephosphorylated to reversibly disassemble the NPC during mitosis. Also of interest is how proteins released from NPCs during mitosis play roles to facilitate mitotic progression and coordinate nuclear restructuring with successful high fidelity DNA segregation.

Osmani Lab Members
Graduate Students:
- Mahesh Chemudupati
- Nandini Shukla
- Subbulakshmi Suresh
Research Staff:
- Aysha Osmani
Undergraduate Students:
- Nichole Bishop
- Alisha Lad
- Andrew Goodall
Current Projects
.jpg) |
Nandini Shukla Analysis of novel proteins acting at the interface between nuclear pore complex associated proteins and the cytoskeleton. |
.jpg) |
Subbulakshmi Suresh Analysis of a new nuclear pore complex protein having dual roles, one at interphase nuclear pores and another at mitotic chromatin. |
.jpg) |
Mahesh Chemudupati
|
.jpg) |
Aysha Osmani
|
Recent Publications
-
Suresh S, Markossian S, Osmani AH, Osmani SA. (2017) Mitotic nuclear pore complex segregation involves Nup2 in Aspergillus nidulans. J Cell Biol. 2017 Jul 26. pii: jcb.201610019. doi: 10.1083/jcb.201610019. [Epub ahead of print]. PMID:28747316
-
De Souza CP, Hashmi SB, Hage N, Fitch RM, Osmani AH, Osmani SA. (2017) Location and functional analysis of the Aspergillus nidulans Aurora kinase confirm mitotic functions and suggest non-mitotic roles. Fungal Genet Biol. 2017 Jun;103:1-15. doi: 0.1016/j.fgb.2017.03.004. Epub 2017 Mar 14. PMID:28315405
-
Shukla N, Osmani AH, Osmani SA. (2017) Microtubules are reversibly depolymerized in response to changing gaseous microenvironments within Aspergillus nidulans biofilms. Mol Biol Cell. 2017 Mar 1;28(5):634-644. doi: 10.1091/mbc.E16-10-0750. Epub 2017 Jan 5. PMID:28057761
-
Chemudupati M, Osmani AH, Osmani SA. (2016) A mitotic nuclear envelope tether for Gle1 also impacts nuclear and nucleolar architecture. Mol Biol Cell. 2016 Sep 14. pii: mbc.E16-07-0544. PMID:27630260
-
Liu HL, Osmani AH, Osmani SA. (2015) The Inner Nuclear Membrane Protein Src1 Is Required for Stable Post-Mitotic Progression into G1 in Aspergillus nidulans. PLoS ONE 10(7): e0132489. doi:10.1371/journal.pone.0132489
-
Markossian S, Suresh S, Osmani AH, Osmani SA. (2015) Nup2 requires a highly divergent partner, NupA, to fulfill functions at nuclear pore complexes and the mitotic chromatin region. Mol. Biol. Cell. 26, 605-621.
-
Govindaraghavan M, Anglin SL, Osmani AH, Osmani SA. (2014) The Set1/COMPASS histone H3 methyltransferase helps regulate mitosis with the CDK1 and NIMA mitotic kinases in Aspergillus nidulans. Genetics. 197, 1225–1236
-
Govindaraghavan M, McGuire Anglin SL, Shen KF, Shukla N, De Souza CP, Osmani SA. (2014) Identification of interphase functions for the NIMA kinase involving microtubules and the ESCRT pathway. PLoS Genet. 10:e1004248. doi: 10.1371/journal.pgen.1004248. PMID:24675878.
-
De Souza CP, Hashmi SB, Osmani AH, Osmani SA. (2014) Application of a new dual localization-affinity purification tag reveals novel aspects of protein kinase biology in Aspergillus nidulans. PLoS One. 9:e90911. doi: 10.1371/journal.pone.0090911. PMID:24599037.
-
Shen KF, Osmani AH, Govindaraghavan M, Osmani SA. (2014) Mitotic regulation of fungal cell-to-cell connectivity through septal pores involves the NIMA kinase. Mol Biol Cell. 25:763-75. PMID:24451264 (Cover article and a Highlights from MBoC Selection).
-
Shen KF, Osmani SA. (2013) Regulation of mitosis by the NIMA kinase involves TINA and its newly discovered partner, An-WDR8, at spindle pole bodies. Mol. Biol. Cell. 24, 3842-3856
-
Larson JR, Facemyer EM, Shen KF, Ukil L, Osmani SA. (2014) Insights into Dynamic Mitotic Chromatin Organization Through the NIMA Kinase Suppressor SonC, a Chromatin-Associated Protein Involved in the DNA Damage Response. Genetics. 196:177-95.
-
Govindaraghavan M, Lad AA, Osmani SA. (2014) The NIMA Kinase Is Required To Execute Stage-Specific Mitotic Functions after Initiation of Mitosis. Eukaryotic Cell. 13, 99-109
-
De Souza CP, Hashmi SB, Osmani AH, Andrews P, Ringelberg CS, Dunlap JC, Osmani SA. (2013) Functional analysis of the Aspergillus nidulans kinome. PLoS One. 8 (3):e58008, 1-27.
-
De Souza CP and Osmani SA. (2011) A new level of spindle assembly checkpoint inactivation that functions without mitotic spindles. Cell Cycle Volume 10 Issue 22
-
De Souza, C.P., S.B. Hashmi, X. Yang, and S.A. Osmani. (2011) Regulated inactivation of the spindle assembly checkpoint without functional mitotic spindles. The EMBO Journal. 30:2648-2661.
-
Markina-Inarrairaegui, A., O. Etxebeste, E. Herrero-Garcia, L. Araujo-Baza, J. Fernandez-Martinez, J.A. Flores, S.A. Osmani, and E.A. Espeso. (2011) Nuclear transporters in a multinucleated organism: functional and localization analyses in Aspergillus nidulans. Molecular Biology of the Cell. 22:3874-86.
-
De Souza CP and Osmani SA. (2010) Mitotic cell cycle control. Cellular and Molecular Biology of Filamentous Fungi. ASM press, pp 63-80.
-
Liu, H.L., A.H. Osmani, L. Ukil, S. Son, S. Markossian, K.F. Shen, M. Govindaraghavan, A. Varadaraj, S.B. Hashmi, C.P. De Souza, and S.A. Osmani. (2010) Single-step affinity purification for fungal proteomics. Eukaryotic cell. 9:831-833.
-
Nayak, T., H. Edgerton-Morgan, T. Horio, Y. Xiong, C.P. De Souza, S.A. Osmani, and B.R. Oakley. (2010) Gamma-tubulin regulates the anaphase-promoting complex/cyclosome during interphase. The Journal of Cell Biology. 190:317-330.
-
Bathe, F., C. Kempf, S.A. Osmani, A.H. Osmani, S. Hettinger, E. Wohlmann, and R. Fischer. (2010) Functional characterization of a new member of the Cdk9 family in Aspergillus nidulans. Eukaryotic Cell. 9:1901-1912.
-
De Souza, C.P., S.B. Hashmi, T. Nayak, B. Oakley, and S.A. Osmani. (2009) Mlp1 acts as a mitotic scaffold to spatially regulate spindle assembly checkpoint proteins in Aspergillus nidulans. Molecular Biology of the Cell. 20:2146-2159.
-
De Souza, C.P., and S.A. Osmani. (2009) Double duty for nuclear proteins--the price of more open forms of mitosis. Trends in Genetics. 25:545-554.
-
Liu, H.L., C.P. De Souza, A.H. Osmani, and S.A. Osmani. (2009) The three fungal transmembrane nuclear pore complex proteins of Aspergillus nidulans are dispensable in the presence of an intact An-Nup84-120 complex. Molecular Biology of the Cell. 20:616-630.
-
Son, S., and S.A. Osmani. (2009) Analysis of all protein phosphatase genes in Aspergillus nidulans identifies a new mitotic regulator, fcp1. Eukaryotic Cell. 8:573-585.
-
Ukil, L., C.P. De Souza, H.L. Liu, and S.A. Osmani. (2009) Nucleolar separation from chromosomes during Aspergillus nidulans mitosis can occur without spindle forces. Molecular Biology of the Cell. 20:2132-2145.
-
Goldman, G. H and S. A. Osmani. Eds. (2008) The Aspergilli: Genomics, Medical Aspects, Biotechnology and Research Methods CRC Press.
-
Wortman, J.R., J.M. Gilsenan, V. Joardar, J. Deegan, J. Clutterbuck, M.R. Andersen, D. Archer, M. Bencina, G. Braus, P. Coutinho, H. von Dohren, J. Doonan, A.J. Driessen, P. Durek, E. Espeso, E. Fekete, M. Flipphi, C.G. Estrada, S. Geysens, G. Goldman, P.W. de Groot, K. Hansen, S.D. Harris, T. Heinekamp, K. Helmstaedt, B. Henrissat, G. Hofmann, T. Homan, T. Horio, H. Horiuchi, S. James, M. Jones, L. Karaffa, Z. Karanyi, M. Kato, N. Keller, D.E. Kelly, J.A. Kiel, J.M. Kim, I.J. van der Klei, F.M. Klis, A. Kovalchuk, N. Krasevec, C.P. Kubicek, B. Liu, A. Maccabe, V. Meyer, P. Mirabito, M. Miskei, M. Mos, J. Mullins, D.R. Nelson, J. Nielsen, B.R. Oakley, S.A. Osmani, T. Pakula, A. Paszewski, I. Paulsen, S. Pilsyk, I. Pocsi, P.J. Punt, A.F. Ram, Q. Ren, X. Robellet, G. Robson, B. Seiboth, P. van Solingen, T. Specht, J. Sun, N. Taheri-Talesh, N. Takeshita, D. Ussery, P.A. vanKuyk, H. Visser, P.J. van de Vondervoort, R.P. de Vries, J. Walton, X. Xiang, Y. Xiong, A.P. Zeng, B.W. Brandt, M.J. Cornell, C.A. van den Hondel, J. Visser, S.G. Oliver, and G. Turner. (2009) The 2008 update of the Aspergillus nidulans genome annotation: a community effort. Fungal Genetics and Biology. 46 Suppl 1:S2-13.
-
Taheri-Talesh, N., T. Horio, L. Araujo-Bazan, X. Dou, E.A. Espeso, M.A. Penalva, S.A. Osmani, and B.R. Oakley. (2008) The tip growth apparatus of Aspergillus nidulans. Molecular Biology of the Cell. 19:1439-1449.
-
Ukil, L., A. Varadaraj, M. Govindaraghavan, H.L. Liu, and S.A. Osmani. (2008) Copy number suppressors of the Aspergillus nidulans nimA1 mitotic kinase display distinctive and highly dynamic cell cycle-regulated locations. Eukaryotic Cell. 7:2087-2099.
-
De Souza, C.P., and S.A. Osmani. (2007) Mitosis, not just open or closed. Eukaryotic Cell. 6:1521-1527.
-
De Souza, C.P., S.B. Hashmi, K.P. Horn, and S.A. Osmani. (2006) A point mutation in the Aspergillus nidulans sonBNup98 nuclear pore complex gene causes conditional DNA damage sensitivity. Genetics. 174:1881-1893.
-
Nayak, T., E. Szewczyk, C.E. Oakley, A. Osmani, L. Ukil, S.L. Murray, M.J. Hynes, S.A. Osmani, and B.R. Oakley. (2006) A versatile and efficient gene-targeting system for Aspergillus nidulans. Genetics. 172:1557-1566.
-
Osmani, A.H., J. Davies, H.L. Liu, A. Nile, and S.A. Osmani. (2006) Systematic deletion and mitotic localization of the nuclear pore complex proteins of Aspergillus nidulans. Molecular Biology of the Cell. 17:4946-4961.
-
Osmani, A.H., B.R. Oakley, and S.A. Osmani. (2006) Identification and analysis of essential Aspergillus nidulans genes using the heterokaryon rescue technique. Nature Protocols. 1:2517-2526.
-
Szewczyk, E., T. Nayak, C.E. Oakley, H. Edgerton, Y. Xiong, N. Taheri-Talesh, S.A. Osmani, and B.R. Oakley. (2006) Fusion PCR and gene targeting in Aspergillus nidulans. Nature Protocols. 1:3111-3120.
